“Biodegradable Block Copolymer–Tannic Acid Glue” by Jongmin Park, Eunsook Park, Siyoung Q. Choi, Jingxian Wu, Jihye Park, Hyeonju Lee, Hyungjun Kim, Haeshin Lee, and Myungeun Seo was published in JACS Au. Jongmin in our group, now a senior researcher at the Korea Research Institute of Chemical Technology (KRICT), and Dr. Eunsook Park in the Haeshin Lee group led the work in collaboration with the Hyungjun Kim group in the department of chemistry, KAIST, and prof. Siyoung Choi in the department of chemical and biomolecular engineering, KAIST.
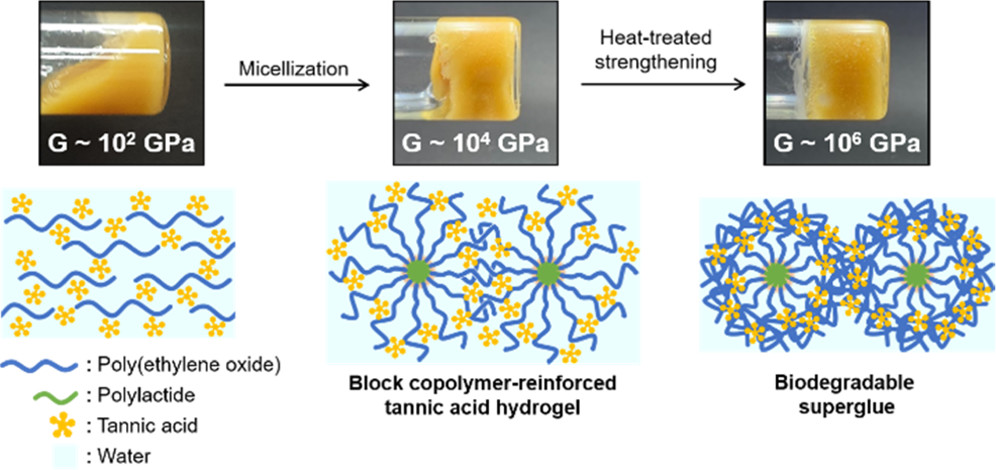
Tannic acid is a natural polyphenol found in fruit peels, nuts, cacao, etc. It contains a glucose unit in the center surrounded by several polygalloyl groups. Numerous hydroxyl groups on the periphery promote adhesion onto other molecules and surfaces, such as protein. The astringent taste of wine comes from the strong binding of tannic acid to a tongue. Tannic acid can also interact with water-soluble polymers in water, such as poly(ethylene oxide) (PEO), resulting in the precipitation of yellow sticky matter called coacervate. The denser coacervate phase, separated from the dilute solution, is a viscoelastic liquid keeping some water. The Lee group pioneered its application as a medical adhesive helping hemostasis (see the article here published in Advanced Functional Materials in 2015). However, being a viscoelastic liquid, the relatively low elastic modulus and weak shear strength limited its use.
We came into play and added a biocompatible, water-insoluble polylactide (PLA) chain to the PEO end to make an amphiphilic diblock copolymer. The copolymer forms micelles composed of PLA core and PEO corona in water. At high concentrations, the micelles start to pack and create elasticity – the solution becomes a viscoelastic solid (i.e., hydrogel) with much higher (can be more than three orders of magnitude high) elastic moduli. As the hydrogen bonding between tannic acid and PEO retains the sticky character, a biocompatible yet much stronger adhesive can be realized.
Interestingly, this hydrogel is strengthened over repeated heating and cooling, somewhat similar to the metal hardening process, and improves its adhesion strength significantly. Heating above the lower critical solution temperature of PEO in water turns the gel into a fluidic sol as the hydrogen bonds weaken. Still, the micellar packing persists and even improves the order. The material also kicks some water out as the polymer chain becomes less hydrophilic and dehydrated. We believe that tannic acid can find more PEO chains as a hydrogen bonding acceptor than water molecules in this condition. The rearranged hydrogen bonds in the well-defined micellar packing enhance the elastic modulus upon cooling and reabsorbing water. While the elasticity boost is also observed with the PEO homopolymer, the effect quickly diminishes, probably because the PEO chain can easily change its conformation and interact with water again (stress relaxation). The computational simulations performed by the Hyungjun Kim group support the hypothesis.
We further demonstrated a preliminary yet rather exciting application of this biocompatible glue for follicle-free hair transplantation. The adhesive seems to firmly hold the end of the hair strand in the tissue when the glued strand is transplanted. While the adhesive may not function as a follicle and help the hair strand grow further, this repeatable and less invasive approach may complement conventional hair transplantation.
The article has hit 1781 views as of October 6, 2022 and been one of the Most Read Articles since its publication. The whole article is available at the following link: https://pubs.acs.org/doi/10.1021/jacsau.2c00241.